About 1% of children with unilateral retinoblastoma and no family history have two normal copies of RB1.
Cells in the tumour and throughout the child’s body show no damage to the RB1 gene. In these children, the cancer is caused by too many copies of the MYCN gene.
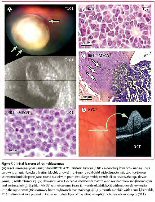
RB1+/+MYCNA retinoblastoma is characterized by unilateral disease, distinct histological features, only a few genomic copy-number changes that define RB1 retinoblastoma, and very early age of diagnosis.
Discovery of RB1+/+MYCNA Retinoblastoma
RB1+/+MYCNA retinoblastoma was first reported in March 2013. Several RB1 genetics researchers found a small but consistent number of children with early onset, highly aggressive retinoblastoma appeared to have two fully intact RB1 genes. They hypothesized that a gene other than RB1 was causing the cancer, and formed an international group to research this theory.
Researchers from Toronto (Canada), Paris (France), Essen (Germany), Amsterdam (Netherlands) and Christchurch (New Zealand) studied tumours from 1068 children with unilateral retinoblastoma and no family history. They compared those with no evidence of RB1 mutations (RB1+/+) with tumours carrying a mutation in both RB1 genes (RB1–/–).
They looked at associated promoter genes (genes that cause cancer and become mutated once RB1 is damaged) and found amplification (more copies than normal) of MYCN (pronounced “Mick-En”) in roughly half the RB1+/+ tumours. MYCN is the gene most associated with high risk neuroblastoma. Subsequent extensive studies confirmed the discovery, and determined this type of retinoblastoma accounts for about 1.4% of unilateral non-familial retinoblastoma.
Differences between RB1-/- and RB1+/+MYCNA Retinoblastoma
RB1+/+MYCNA tumours have two normal copies of RB1, whereas both RB1 genes are mutated in classic RB1 initiated retinoblastoma. They also have fewer changes in genes characteristic of retinoblastoma than do RB1–/– tumours.
Median age at diagnosis of RB1+/+MYCNA tumours is 4·5 months, compared with 24 months for non-familial unilateral RB1–/– retinoblastoma.
RB1+/+MYCNA retinoblastomas are already large in very young children. In children of equivalent age with an RB1 mutation, tumours are usually much smaller.
Under the microscope after enucleation, RB1+/+MYCNA retinoblastomas resemble high risk, poor outcome neuroblastoma. However, they arise from immature retinal cells, and are therefore classified as retinoblastoma.
Treatment Implications
Diagnosis of unilateral retinoblastoma in the first year of life is most often considered an indication of heritable RB1 mutation. Due to the risk of cancer developing in the other eye in children with heritable RB1 mutation, greater effort is invested to save the affected eye.
However, RB1+/+MYCNA retinoblastoma is highly aggressive and usually advanced at diagnosis, with potential for cancer to already be threatening the baby’s life. Attempting to save an eye with RB1+/+MYCNA cancer may be very dangerous.
Risk for RB1+/+MYCNA in a 6 month old baby with unilateral retinoblastoma and no family history is 20%. Risk increases with younger age at diagnosis. Considering age and size of the unilateral tumour at diagnosis, it is possible to predict clinically the child’s risk for RB1+/+MYCNA retinoblastoma. Though these tumours display features of high risk neuroblastoma, prompt removal of the eye and careful pathology to guide postoperative care can ensure excellent outcome for the child.
Clinical Features of Retinoblastoma
Figure reprinted from The Lancet Oncology, Vol. 14. Rushlow, D, et al. Characterisation of retinoblastomas without RB1 mutations: genomic, gene expression, and clinical studies. Epub ahead of print. © (2013), with permission from Elsevier.